Tel: +86-29-88193552, 88193551
Fax: +86-29-88193353
E-Mail: sales@dashengpharm.com
URL: www.dashengpharm.com
Chlorhexidine Hydrochloride (GMP)
1. CAS RN: 3697-42-5
2.English Name:Chlorhexidine Hydrochloride
Chemical Name:Synonyms:Chlorhexidine Hydrochloride; 2,4,11,13-Tetraazatetradecanediimidamide, N,N'-bis(4-chlorophenyl)-3,12-diimino-, dihydrochloride; Chlorhexidine dihydrochloride; 1,1'-Hexamethylenebis(5-(p-chlorophenyl)biguanide) dihydrochloride; Biguanide, 1,1'-hexamethylenebis(5-(p-chlorophenyl)-, dihydrochloride; 2,2'-hexane-1,6-diylbis(1-{amino[(4-chlorophenyl)amino]methylidene}guanidine) dihydrochloride
3.Structural Formula:
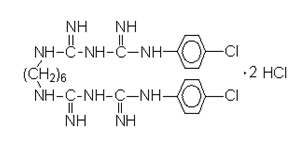
4. Molecular formula & molecular weight: C22H30Cl2N10.2HCl 578.37
5. Characters: It’s a white or almost white crystal powder, odorless, bitter taste, hardly soluble in water or alcohol. Melting point: 255~259℃, decompounding after fusing.
6. Quality Standard: EP7.0
7. Usage: It is a disinfect and antiseptic medicine, strong function of broad-spectrum bacteriostasis, sterilization, used for disinfecting hands, medical apparatus and instruments, wound ,skin and so on.
8. Storage: It should be kept in cool, dark and dry place, stored in sealed containers.
9. Packing: paper bucket, 20kg/drum,
10. HS CODE: 2921 4200.90


 MSN Messanger
MSN Messanger
